What are smart sensors and digital biomarkers?
Smart sensors are devices that take input from their environments and process that data before transmitting it. They have been used in a number of applications, including in the mobile and portable device space (think phones and tablets). In order to accomplish their tasks, smart sensors are equipped with signal conditioning, embedded algorithms, and a digital interface. The most significant feature of a smart sensor is its ability to communicate, which can be done by displaying the data directly to the user or transferring wirelessly.
The term “digital biomarkers” refers to data that are directly collected about health or disease management through digital health technologies to explain, influence, and/or predict health-related results. They span a broad range of diagnostic and prognostic measurements that are frequently made outside of the clinical environment using home-based connected products including wearable, implantable, and ingestible devices and sensors.
Digital biomarker applications:
Smart sensor technology has a wide range of applications in healthcare, with devices built to record data including blood pressure, skin conductance, mobility, posture, oxygen levels, respiration, sleep, temperature, and heart rate. In recent years, gadgets such as Fitbit, Misfit, Jawbone, Apple Health, Sleep as Android, WIWE, MocaCare, and Skeeper — in other words, fitness trackers, step counters, health apps, sleep sensors, pocket EKGs, and blood pressure or other health parameter measuring devices — have become significant players in the health, wellness, and fitness market. These devices generate an astounding amount of data for individuals monitoring their own personal wellness data and, increasingly, for patients under direct physician supervision, including as part of clinical trials.
What are some examples of best practice cases?
Experience using digital biomarkers as outcome measures in clinical trials is limited but growing fast. Recent years have seen applications of digital biomarkers in different therapeutic areas, including cardiovascular disease, sleep, respiratory conditions, diabetes, and neuropsychiatric diseases.
Cardiovascular diseases
For example, several companies have focused on detecting atrial fibrillation (afib) for cardiovascular disease. While the FDA has not yet approved any algorithms based solely on smartwatch data, a digital biomarker for afib detection is available in conjunction with an approved EKG. Likewise, a trial using nitrate to treat congestive heart failure recently published in The New England Journal of Medicine used an accelerometer as the primary outcome measure.
Sleep
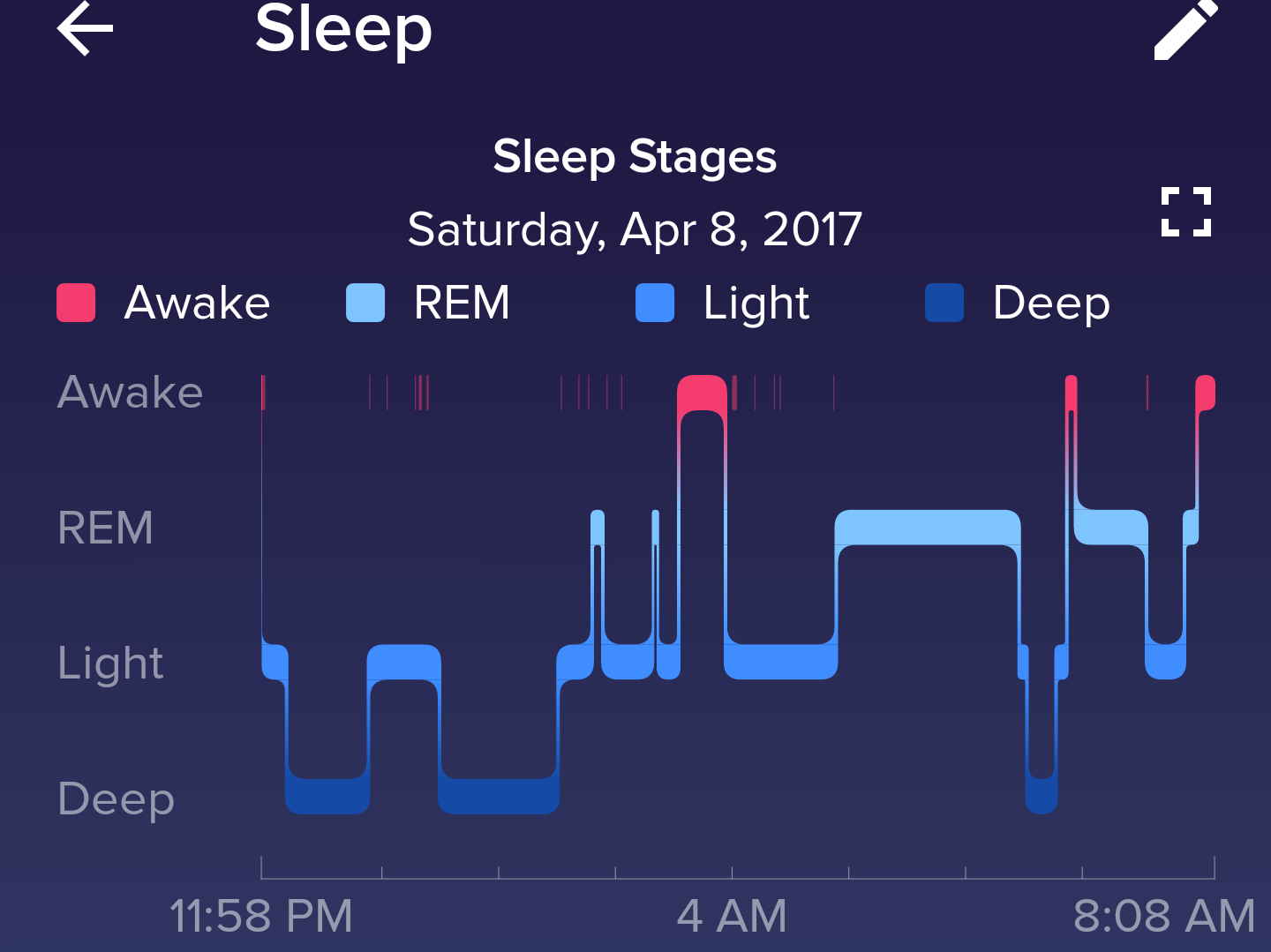
As for sleep, the road to commercial use of digital biomarkers seems to be very different, as it includes a healthy living space as well. Because much less validation is required, a variety of digital biomarkers such as Fitbit’s sleep stages feature are commercially available. Fitbit has confirmed the ability of wrist-worn trackers that incorporate movement and cardiac sensors to accurately determine light, deep, and REM sleep stages. Scientists presented the findings of the 2017 study “Estimation of Sleep Stages Using Cardiac and Accelerometer Data from a Wrist-Worn Device” in the journal Sleep. The results of Fitbit’s study demonstrate that these devices can be used to track sleep stages with a reasonable degree of accuracy in normal adult sleepers, according to Fitbit. Having the ability to collect reliable sleep stage data on wrist-worn devices can help simplify sleep research and increase public awareness of sleep issues.
Respiratory disorders
Through its sensor-outfitted inhalers, Propeller Health is able to identify environmental triggers for patients with asthma, chronic obstructive pulmonary disorder, and other respiratory conditions. The Propeller Health system includes a sensor that attaches to a rescue inhaler or to controller inhaler medications for patients to better manage their conditions. This system is FDA-approved as a medical device. In addition, the company has also launched the first predictive tool for asthma risk based on environment.

Diabetes
Diabetes remains a highly innovative field for pharmaceutical companies in terms of digital therapeutics. For example, San Francisco–based Omada Health is a pioneer in bringing evidence-based digital therapeutics to the area of prediabetes and diabetes. Omada offers special online coaching programs to help prevent chronic health problems such as diabetes. The company’s first product, Prevention, is an online version of the Diabetes Prevention Program, a series of interventions by the National Institutes of Health (NIH) based on studies of how diet and exercise can reduce patients’ chances of developing diabetes.
The company aims to incorporate the best in behavioral psychology and provides wireless monitoring devices that allow Omada coaches to work closely with patients. Each participant is given a wireless digital scale and a pedometer. They then use an online platform to interact with personal health coaches for daily feedback. The results have been positive so far. On average, participants lost 4.7% more body weight than an average weight loss of 2.4% for personal diabetes prevention programs.

Recently, Merck and Amazon announced that they are exploring the creation of voice-based diabetes tools. In Merck’s new partnership with Amazon Web Services, the company is aiming to develop digital voice-enabled solutions for people with diabetes using the Amazon Echo device and its well-known voice-enabled assistant, Alexa.
Neuropsychiatric disease
With regard to neuropsychiatric diseases, much research is currently in progress. Of note, researchers have begun examining elderly adults with dementia and patients with Parkinson’s and Huntington’s diseases through wearables, accelerometers, and smartwatch devices. Efforts to reduce the severity of conditions such as Parkinson’s disease, multiple sclerosis, and Alzheimer’s disease by effectively treating symptoms are benefiting from the application of digital biomarkers.
Roche developed a Parkinson’s disease app to capture voice-related information with the aim of capturing data that may be associated with early symptoms of Parkinson’s disease. The application can collect voice information while people talk on the phone and measure stability when users hold their devices.
Neurotrack remotely measures cognitive ability to assess cognition in patients with Alzheimer’s disease. It is a digital test for detecting cognitive deficits using safe, noninvasive technology and brain health assessment. This online test has the potential to be an inexpensive and easy-to-use approach to detecting the state of Alzheimer’s before the development of clinical symptoms. It assesses what researchers call “recognition memory,” which depends on the integrity of the brain’s hippocampal region. The 5-minute assessment measures the health of the hippocampus using eye-tracking technology. These measurements are used to predict cognitive decline before symptoms appear. Assessments are designed to be taken repeatedly so that users can track how their memory changes over time. After each assessment, users receive a memory score and recommendations for how to improve their brain health.
Biogen partnered with PatientsLikeMe to understand physical activity measurements in patients with multiple sclerosis.
How can smart sensors and digital biomarkers empower the pharma industry’s existing treatments?
Failure is a common situation in drug development, especially for neurodegenerative diseases. Compared to other therapeutic areas, drug development for neurological disorders usually takes longer, costs more, and is less likely to be successful. Such failures are costly, discouraging future investments and hindering the development of treatments. Since success is rare, there remains a greater unmet medical need in this area. Given the lack of reliable physiological biomarkers, digital biomarkers may help to address current shortcomings. Objective, high-frequency data can guide critical decision-making in therapeutic development and enable a more effective assessment of the treatment of increasingly common diseases. Digital biomarkers such as mobile and wearable technologies (smart phones, tablets, watches, rings, and suits) present a unique opportunity to massively detect neurodegenerative disorders in a timely and economical fashion due to:
- Widespread use of such technologies
- Immediate access to information due to inherent connectivity
- Increasing sensitivity and plurality of onboard sensors
- The nature of sensors that are uniquely equipped to study such physical and cognitive abilities or symptoms
- The extremely low burden on the healthcare system, since these devices are increasingly in use by large segments of the population
Because of these advantages, digital biomarkers such as mobile and wearable devices can empower existing treatments and aid in the development of novel approaches.
Conclusion
Recently, pharmaceutical companies have recognized the tremendous potential of digital biomarkers, and a plethora of studies are underway to better understand health and disease through the lens of these data. Furthermore, researchers will be able to better analyze the causes of diseases through digital biomarkers and to predict future health outcomes. Widespread measurements of digital biomarkers, as enabled by user-friendly devices equipped with smart sensors, will help move healthcare forward from a more reactive approach to a more preventive approach.
It should be noted that the use of digital biomarkers as part of drug development or healthcare delivery requires years of careful research and validation, which in most cases is still underway or just poised to begin. Moreover, the vast majority of digital biomarkers have yet to find practical applications. Although digital biomarkers have not yet been approved as a drug development tool, biopharmaceutical companies are working to develop new endpoints based on digital biomarkers. As a result, research on digital biomarkers will increase rapidly in the coming years.








